Ctti-Clinicaltrials.org recives any estimated n/a unique visitors and n/a unique page views per day. Revenue gained from these much visits may be n/a per day from various advertising sources. The estimated worth of site is n/a.
- Website Age
n/a
- Alexa Rank no-data
- Country
 United States
United States
- IP Address
152.16.0.233
HTML SIZE INFORMATION
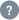
Text / Code Ratio
13.99 %
ctti-clinicaltrials.org has a website text/code ratio of 13.99 %. Search engine crawlers tend to not pick up pages with inadequate content.
IMPORTANT HTML TAGS AND COUNTS

- H10
- H21
- H30
- H49
- H515
- H611
H2
| No |
Text |
| 1 |
Mıssıon: to develop and drive adoption of practices that will increase the quality and efficiency of clinical trials |
H4
| No |
Text |
| 1 |
Who we are |
| 2 |
What we do |
| 3 |
Briefing room |
| 4 |
Take action |
| 5 |
Database updated |
| 6 |
Resources available |
| 7 |
Report now avaılable |
| 8 |
New rwd recommendations |
| 9 |
New tool available |
H5
| No |
Text |
| 1 |
Projects (2) |
| 2 |
Programs of related projects (2) |
| 3 |
Upcoming events |
| 4 |
Recommendations |
| 5 |
Tools |
| 6 |
Publications |
| 7 |
New cttı project aims to ıncrease ınclusion of minority pop****tions in clinical trials |
| 8 |
Cttı paper discusses development of patient group engagement tool to benefit clinical trials |
| 9 |
Cttı paper discusses suitability of patient registries for embedded clinical trials |
| 10 |
Cttı feasibility studies database adds new resources |
| 11 |
Recording now available: strategies for engaging racial and ethnic minority patient pop****tions in covıd-19 trials |
| 12 |
Web conference recordings available: listen to stakeholder engagement on ıch e6 guideline for good clinical practice |
| 13 |
Share your experiences: recognizing the need for diversity in covıd-19 clinical trials |
H6
| No |
Text |
| 1 |
Antibiotic drug development |
| 2 |
Digital health trials |
| 3 |
Article (7) |
| 4 |
Program: antibacterial drug development |
| 5 |
Program: digital health trials |
Text Styling

- STRONG4
- B0
- EM1
- I0
- U0
- CITE3
STRONG
| No |
Text |
| 1 |
CTTI Policy Impact |
| 2 |
Adoption of CTTI's QbD Framework |
| 3 |
Adoption of Central IRB's |
| 4 |
Disclaimer: |
EM
| No |
Text |
| 1 |
Copyright © CTTI 2020. All Rights Reserved |
CITE
| No |
Text |
| 1 |
CTTI Policy Impact |
| 2 |
Adoption of CTTI's QbD Framework |
| 3 |
Adoption of Central IRB's |
LINK ANALYSIS
Total Link Count: 149
Internal Link Count
: 140 
| No |
Text |
Type |
| 1 |
n a v |
text |
| 2 |
Who We Are |
text |
| 3 |
What We Do |
text |
| 4 |
Briefing Room |
text |
| 5 |
Take Action |
text |
| 6 |
Contact Us |
text |
| 7 |
Login |
text |
| 8 |
- |
empty |
| 9 |
Who We Are |
text |
| 10 |
Strategic Plan |
text |
| 11 |
Organizational Structure |
text |
| 12 |
Executive Committee |
text |
| 13 |
Staff |
text |
| 14 |
Steering Committee |
text |
| 15 |
Members***p |
text |
| 16 |
Annual Members***p Fees |
text |
| 17 |
Become A Member |
text |
| 18 |
Become a Patient/Caregiver Rep |
text |
| 19 |
Benefits & Responsibilities |
text |
| 20 |
Funding |
text |
| 21 |
Annual Reports |
text |
| 22 |
What We Do |
text |
| 23 |
Recommendations |
text |
| 24 |
Implementation Tools |
text |
| 25 |
Projects |
text |
| 26 |
COVID-19 |
text |
| 27 |
Data Monitoring Committees (DMCs) |
text |
| 28 |
Electronic Healthcare Data |
text |
| 29 |
Increasing Diversity in Clinical Trials |
text |
| 30 |
Informed Consent |
text |
| 31 |
Informing the Update of ICH E6 |
text |
| 32 |
Investigator Community |
text |
| 33 |
Investigator Qualification |
text |
| 34 |
Large Simple Trials |
text |
| 35 |
Long-Term Opioid Data |
text |
| 36 |
Master Protocol Studies |
text |
| 37 |
Patient Engagement Collaborative (PEC) |
text |
| 38 |
Patient Groups & Clinical Trials |
text |
| 39 |
Pregnancy Testing |
text |
| 40 |
Quality by Design |
text |
| 41 |
Real-World Data |
text |
| 42 |
Recruitment |
text |
| 43 |
Registry Trials |
text |
| 44 |
Safety Reporting |
text |
| 45 |
Single IRB |
text |
| 46 |
Site Metrics |
text |
| 47 |
State of Clinical Trials |
text |
| 48 |
Programs of Related Projects |
text |
| 49 |
Antibiotic Drug Development |
text |
| 50 |
HABP/VABP Studies |
text |
External Link Count
: 9 
| No |
Text |
Type |
| 1 |
Join Our Newsletter |
text |
| 2 |
NIH draft Policy |
text |
| 3 |
FDA’s newest draft guidance on IND safety reporting |
text |
| 4 |
FDA's Guidance on Risk-Based Monitoring |
text |
| 5 |
EMA Reflection Paper |
text |
| 6 |
21st Century Cures |
text |
| 7 |
- |
empty |
| 8 |
- |
empty |
| 9 |
Web Design by NMC |
text |
Nofollow Link Count
: 0
Title Link Count
: 0
WEBSITE SERVER INFORMATION
- Service Provider (ISP)
- Duke University
- Hosted IP Address
- 152.16.0.233
- Hosted Country
 United States
United States- Host Region
- North Carolina , Durham
- Latitude and Longitude
- 35.994 : -78.8986
WEBSITES USING THE SAME IP ADDRESS
WEBSITES USING THE SAME C CLASS IP


 United States
United States

 External Link Count
: 9
External Link Count
: 9 
 152.16.0.233
152.16.0.233 152.16.0.235
152.16.0.235